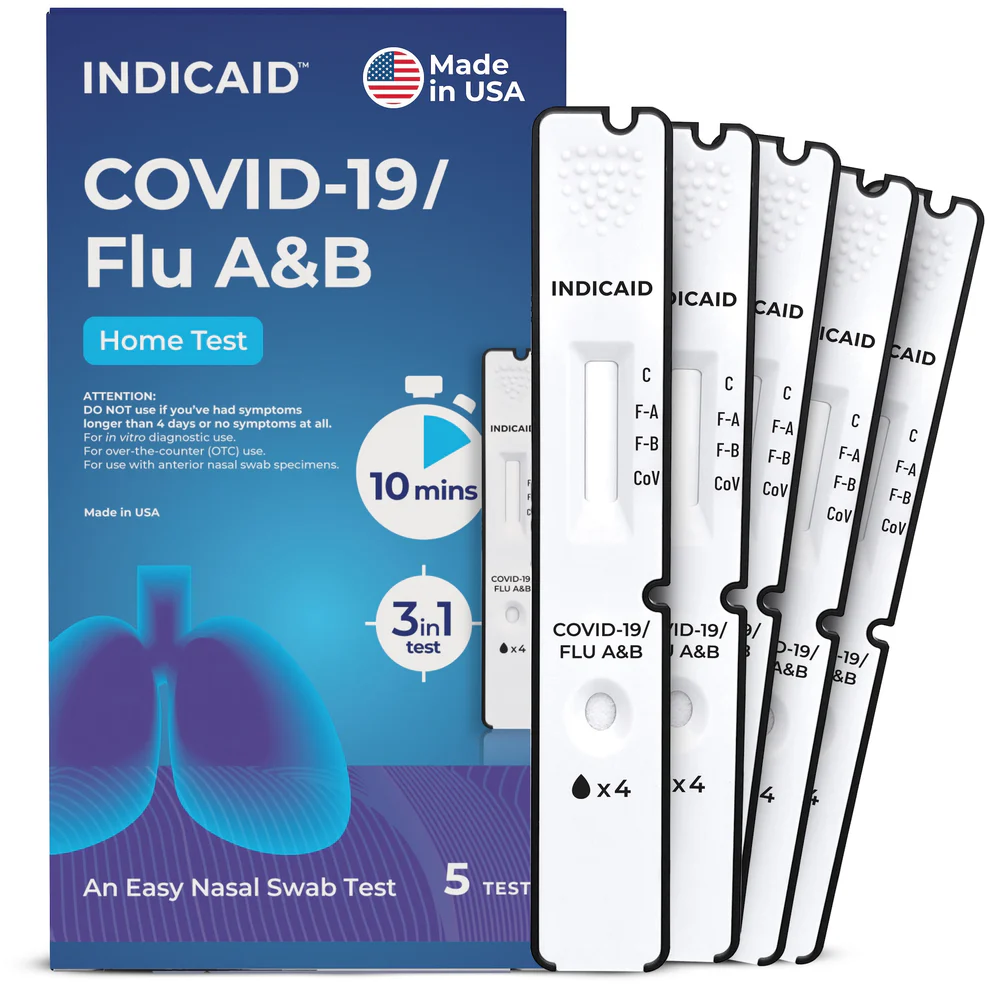
Description
![]()
Contents: (1) per test of – Sealed Test Cassette, Buffer Tube, Swab, Tube Holder (on Box), Quick Reference Instructions
Regulatory Status: FDA 510K Approved
Determinations: Detection of the COVID-19 (SARS-CoV-2), Influenza A, and Influenza B
Processing Time: 10 Minutes
The INDICAID COVID-19 / Flu A&B test is an FDA-cleared, over-the- counter rapid antigen test that delivers three answers with just one swab. Designed for families, travelers, workplaces, and schools—no appointment, no waiting, no lab required.
- Fast: Quickly delivers peace of mind to act with confidence
- Reliable: Clinically validated with FDA 510(k) clearance
- Easy to use: Test at home with no medical training required
- Retail ready: Compact packaging and extended shelf life
Product Resources: