Description
![]()
![]()
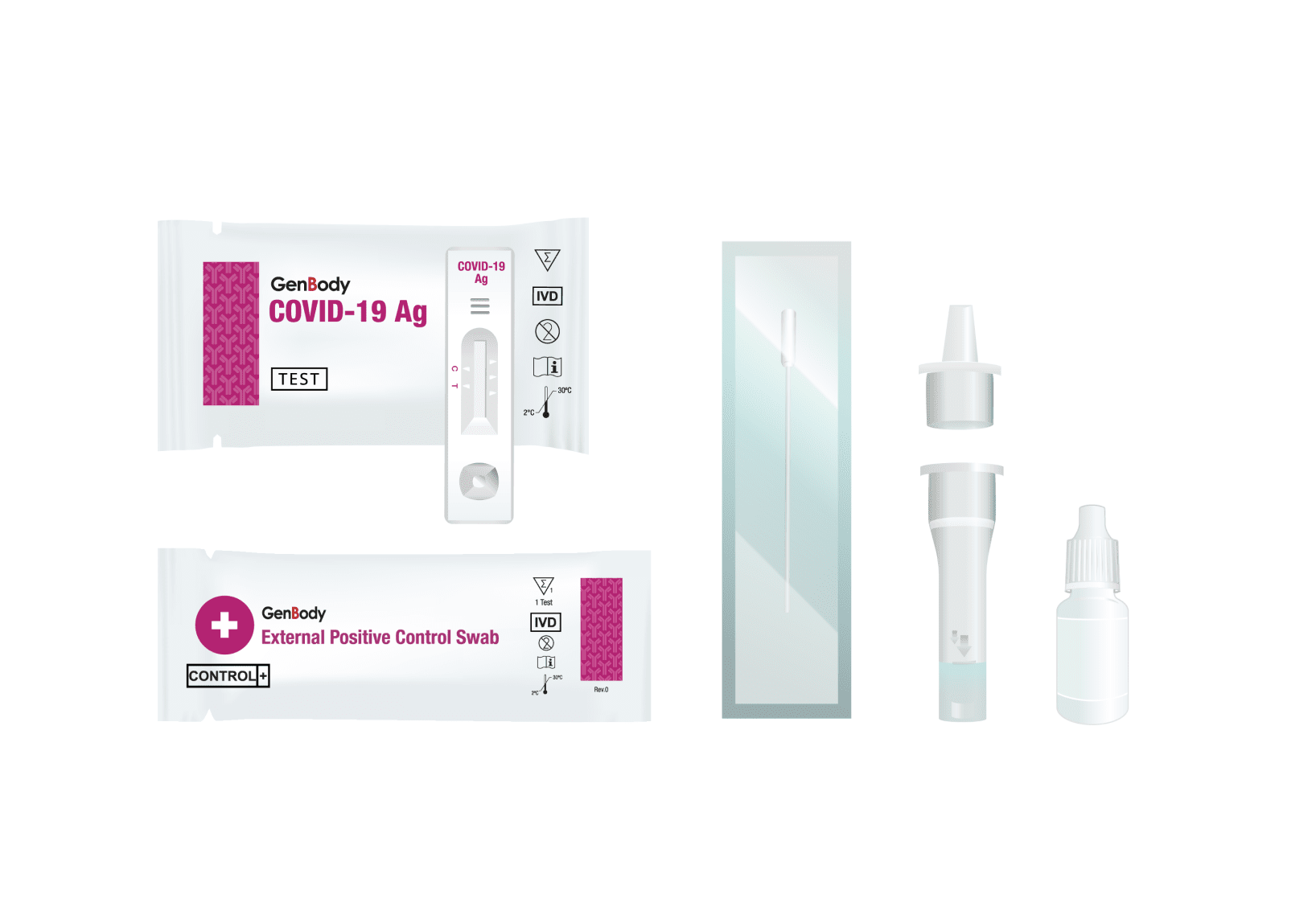
Contents: Test device (25), Pre-Filled Extraction Tubes (25) , Dropper Tips (25), Swabs (25), Positive control swab (1), Negative control swab (1) each, Instructions for use (1) Quick Reference Instructions (QRI) (1)
Regulatory Status: FDA EUA Authorized, CLIA Waived
Determinations: Detection of the SARS-CoV-2 nucleocapsid protein antigen
Processing Time: 15 Minutes
Backed by the NIH, the GenBody Covid-19 Antigen Test, The GenBody COVID-19 Ag test is a rapid, qualitative immunochromatographic assay for the determination of the presence of SARS-CoV-2 antigens in human nasopharyngeal or anterior nasal swab specimens. The test strip in each device contains mouse monoclonal antibodies to the nucleocapsid protein (NP) of SARS-CoV-2.
Features:
- Identifies acute infection with a 92.31% Sensitivity and 99.04% Specificity
- For use in patient care settings
Product Resources:
- CPT Code: 87811QW
- GenBody FDA EUA Letter
- Instructions for Use
- GenBody Package Insert
- Provider Fact Sheet
- Patient Fact Sheet
- GenBody Competitive Advantages
Videos:
- Instructional Video: https://vimeo.com/ 668806493
- Controls: https://vimeo.com/668810632
- Training & Unboxing Video: https://vimeo.com/683509120
Regulatory Status: FDA EUA Authorized – this test has not been FDA cleared or approved but has been authorized by the FDA under an EUA for use by authorized laboratories. CLIA WAIVED
Contents: Test device (25), Pre-Filled Extraction Tubes (25) , Dropper Tips (25), Swabs (25), Positive control swab (1), Negative control swab (1) each, Instructions for use (1) Quick Reference Instructions (QRI) (1)
Determinations: Detection of the SARS-CoV-2 nucleocapsid protein antigen
Storage Requirements: 2° – 30° C
Processing Time: Approximately 15 Minutes
CPT Code: 87811QW
Backed by the NIH, the GenBody Covid-19 Antigen Test, The GenBody COVID-19 Ag test is a rapid, qualitative immunochromatographic assay for the determination of the presence of SARS-CoV-2 antigens in human nasopharyngeal or anterior nasal swab specimens. The test strip in each device contains mouse monoclonal antibodies to the nucleocapsid protein (NP) of SARS-CoV-2.
Highlights include:
- Detects SARS-CoV-2 nucleocapsid protein antigen
- Rapid results in 15-20 minutes
- Identifies acute infection with a 92.31% Sensitivity and 99.04% Specificity
- For use in patient care settings operating under a
- CLIA Certificate of Waiver, Certificate of Compliance or Certificate of Accreditation
The GenBody COVID-19 AG is intended for use by medical professionals or operators trained in performing tests
in point of care settings. The GenBody COVID-19 AG is only for use under the Food and Drug Administration’s
Emergency Use Authorization.
The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
See product links above for additional information or contact your local Stat Technologies sales representative