Description
![]()
![]()
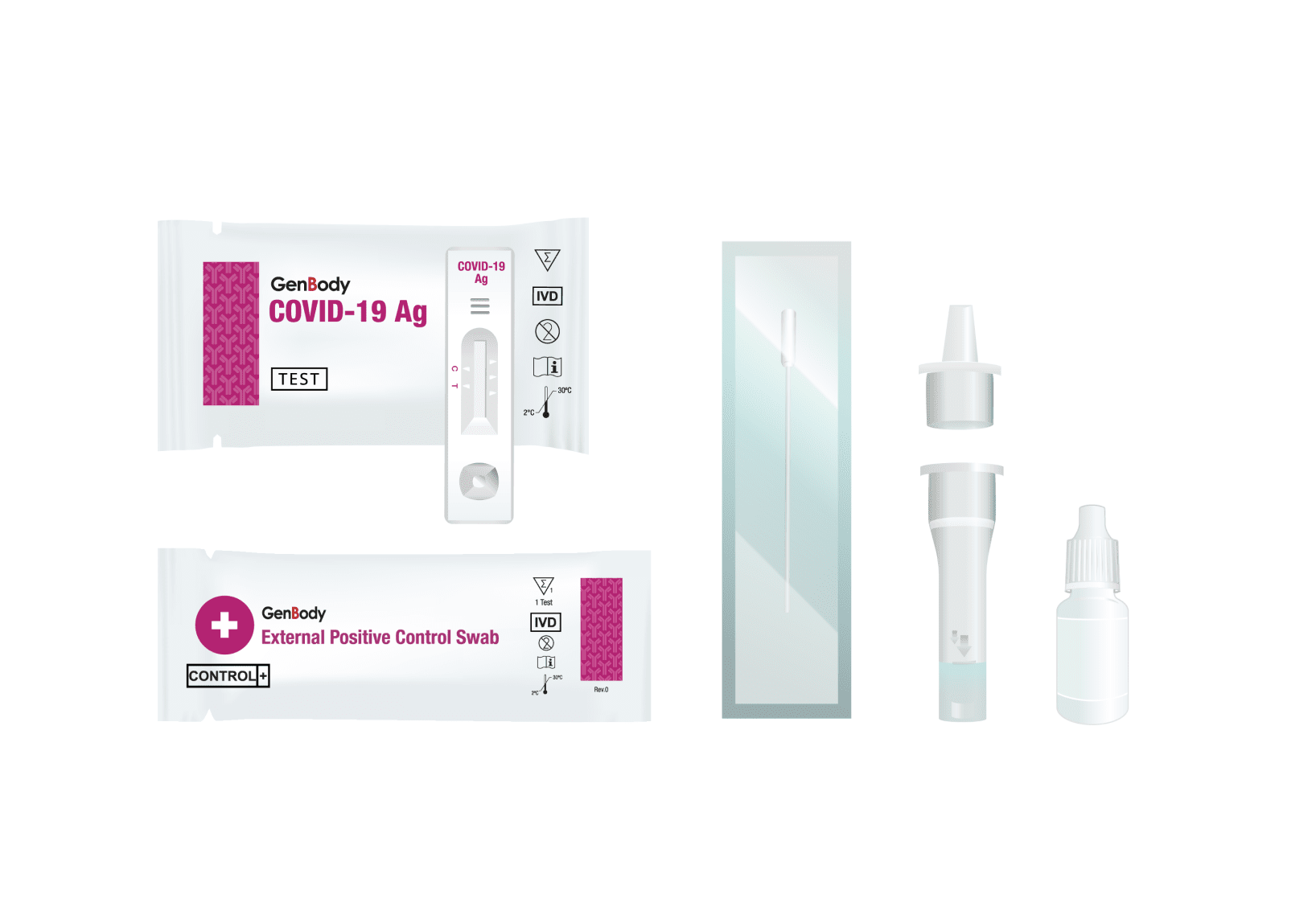
Contents: Test device (25), Pre-Filled Extraction Tubes (25) , Dropper Tips (25), Swabs (25), Positive control swab (1), Negative control swab (1) each, Instructions for use (1) Quick Reference Instructions (QRI) (1)
Regulatory Status: FDA EUA Authorized, CLIA Waived
Determinations: Detection of the SARS-CoV-2 nucleocapsid protein antigen
Processing Time: 15 Minutes
Backed by the NIH, the GenBody Covid-19 Antigen Test, The GenBody COVID-19 Ag test is a rapid, qualitative immunochromatographic assay for the determination of the presence of SARS-CoV-2 antigens in human nasopharyngeal or anterior nasal swab specimens. The test strip in each device contains mouse monoclonal antibodies to the nucleocapsid protein (NP) of SARS-CoV-2.
Features:
- Identifies acute infection with a 92.31% Sensitivity and 99.04% Specificity
- For use in patient care settings
Product Resources:
- CPT Code: 87811QW
- GenBody FDA EUA Letter
- Instructions for Use
- GenBody Package Insert
- Provider Fact Sheet
- Patient Fact Sheet
- GenBody Competitive Advantages
Videos:
- Instructional Video: https://vimeo.com/ 668806493
- Controls: https://vimeo.com/668810632
- Training & Unboxing Video: https://vimeo.com/683509120