Description
![]()
![]()
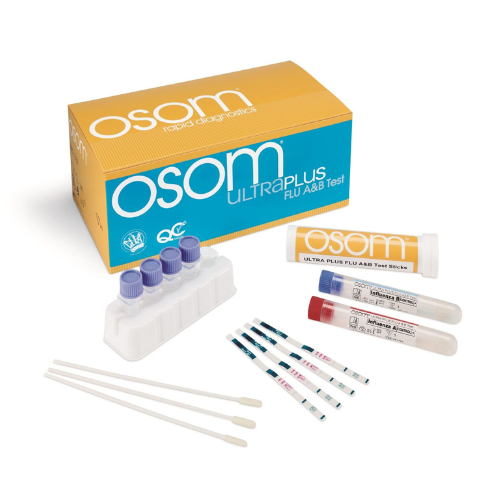
Contents: (25) Test devices + (2) extra for external QC, (25) extraction reagent capsules, (25) sterile swabs, (1) positive control swab, (1) negative control swab, (1) package insert with test procedure page
Regulatory Status: CLIA Waived
Determinations: Detects influenza type A and type B nucleoprotein antigens
Processing Time: 10 Minutes
The OSOM Ultra Flu A&B Test is an in vitro rapid qualitative test that detects influenza type A and type B nucleoprotein antigens directly from a nasal swab obtained from patients with signs and symptoms of respiratory infection.
Product Resources: