Description
![]()
![]()
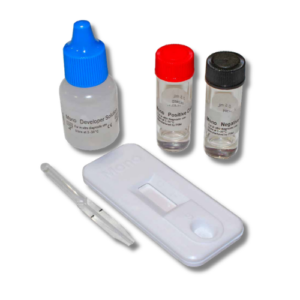
Contents: (25) Test devices, (25) Transfer Pipettes, (1) Developer Solution, (1) Mono Positive Control, (1) Mono Negative Control, (1) Package Insert, (1) Procedure Card
Regulatory Status: CLIA Waived
Determinations: Qualitative Infectious Mononucleosis
Processing Time: 8 Minutes
Status Mono one-step antibody test for IM uses direct solid-phase immunoassay technology for the qualitative detection of IM heterophile antibodies in human serum, plasma or whole blood. In the test procedure, 10 µl serum or plasma are added in the Sample Well (S) located below the result window. For finger-tip or whole blood, 25 µl of blood are collected in
Product Resources: