Description
![]()
![]()
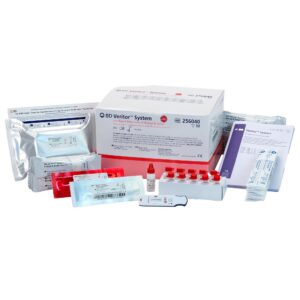
Contents: (25) Test devices, (25) Transfer Pipettes, (1) Developer Solution, (1) Mono Positive Control, (1) Mono Negative Control, (1) Package Insert, (1) Procedure Card
Regulatory Status: FDA Emergency Use Authorization, OTC
Determinations: Detection of the SARS-CoV-2 nucleocapsid protein antigen
Processing Time: 15 Minutes
For at home use, workplace or clinics. Nasal swab- for easy and comfortable self-specimen collection