Description
![]()
![]()
Unit: 1 Box
Regulatory Status: CLIA Waived (whole blood) / Moderately Complex (serum / plasma) / Professional Use Only
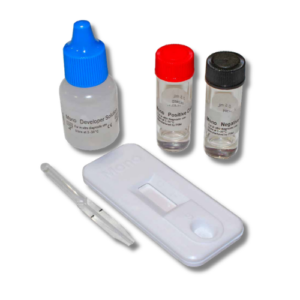
Contents: (1) vial Positive control solution, (1) vial negative control solution, (1) package insert
Determinations: Qualitative Infectious Mononucleosis
Storage Requirements: 36° – 86° F
Processing Time: 8 Minutes
Status Mono Controls Package Insert
Status Mono Controls
Good laboratory practice recommends the periodic use of external control materials to ensure proper kit performance. Using the positive or negative external controls in place of a patient sample, dispense 1 drop of control
solution into the upper end of the sample well(s) of the device.
There are also two internal control features in Status Mono test. A colored control band will always appear at the Control position (C) if the test has been performed correctly and if the device is working properly. This is considered an internal positive procedural control. A clear background in the result window is considered an internal negative procedural control. If the test has been performed correctly and Status Mono device is working properly, the background in the result window will be clear, providing a distinct result.
About Infectious Mononucleosis
Infectious Mononucleosis is caused by the Epstein-Barr virus (EBV), a member of the Herpesviridae family, genus Lymphocryptovirus. The Epstein-Barr virus is a double-stranded, linear, DNA virus that infects the squamous epithelial cells and B lymphocytes in the immune system. Once infected, the individual becomes a lifelong carrier of a low grade infection which is kept in check by the body’s immune defenses. It is estimated that 90% to 95% of the adult population has been exposed to the EBV infection, though not all individuals experience symptoms of the illness. Infectious Mononucleosis is commonly referred to as “Mono” or “the kissing disease” since it is primary transmitted through saliva. EBV has also been found to spread through blood and genital secretions as well.
Virus replication begins in the epithelial cells in the pharynx, causing pharyngitis. The infection then moves to the B lymphocytes, triggering cytotoxic T cells to react against the EBV-infected B cells, producing lymphadenopathy (enlargement of the lymph nodes). These symptoms are often accompanied by fever, malaise, headaches, stiff neck, sweating, chills, rash, and abdominal discomfort and usually develop 4 to 6 weeks after initial exposure. Rare but serious complications may occur that require urgent evaluation or hospitalization. These include tonsillitis, jaundice, anemia, seizures, difficulty breathing or swallowing due to pharyngeal and tracheal obstruction, splenomegaly (leading to possible splenic rupture), meningitis, encephalitis, and Guillain-Barre syndrome.
There is no cure for Infectious Mononucleosis. Mono is characteristically a benign, self-limiting disease which resolves without medical support. Symptoms gradually diminish but may linger for weeks or even months. Supportive care is recommended, such as limiting activity (avoid strenuous exercise and contact sports), getting plenty of rest, hydration, throat lozenges, and over-the-counter medications to relieve symptoms.
The Mononucleosis infection is clinically similar to other illnesses such as acute cytomegalovirus infection, streptococcal pharyngitis, and Toxoplasma gondii. Diagnosis by a health-care provider is important to insure that the illness is not caused by other medical conditions that could require more extensive medical attention and treatment.