Description
![]()
![]()
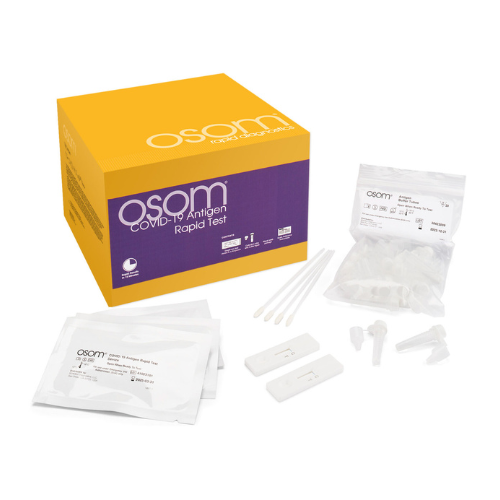
Contents: (40) Test devices, (2×20) Nasal swab specimen collection & dispensing tube containing OSOM Antigen Buffer, (40) sterile swabs, (1) package insert with test procedure page
Regulatory Status: FDA EUA Authorized, CLIA Waived
Determinations: Detection of the SARS-CoV-2 nucleocapsid protein antigen
Processing Time: 15 Minutes
The OSOM® COVID-19 Antigen Rapid Test is a lateral flow chromatographic immunoassay intended for the qualitative detection of the SARS-CoV-2 nucleocapsid protein antigen in direct mid-turbinate (MT) nasal swab specimens collected by a healthcare provider from individuals suspected of COVID-19 within the first 7 days of symptom onset.
Product Resources: