Description
![]()
![]()
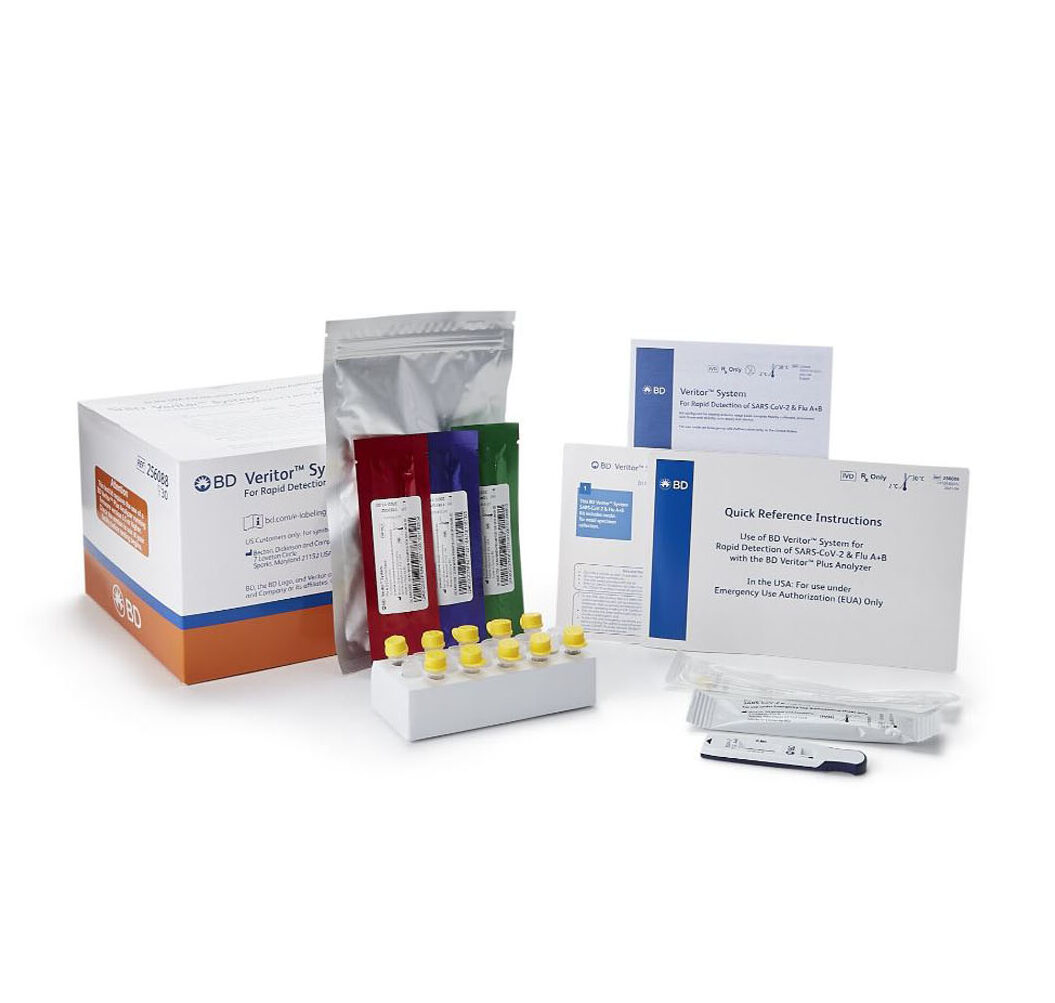
Contents: (30) BD Veritor™ System SARS-CoV-2 & Flu A+B combined tests, (30)sample collection swabs, BD Veritor™ System Extraction Reagent, tube trays, Positive control swabs (SARS-CoV-2, Flu A, Flu B)
Regulatory Status: FDA Emergency Use Authorized- CLIA Waived / Professional Use Only
Determinations: Detection of the SARS-CoV-2 nucleocapsid protein antigen
Processing Time: 15 Minutes
Simultaneous qualitative detection and differentiation of SARS-CoV-2 nucleocapsid antigen and/or influenza A and B nucleoprotein antigens directly from anterior nasal swab samples taken from individuals who are suspected of a viral respiratory infection consistent with COVID-19 by a healthcare provider, within the first six days of symptom onset. Emergency use of this test is limited to authorized laboratories.
Product Resources:
- Covid FLU A+B Package Insert
- Covid Flu A+B Patient Fact Sheet
- Covid Flu A+B Provider Fact Sheet
- FDA EUA Letter
The BD Veritor™ Plus System is a handheld device that enables testing for Flu A+B, Group A Strep, and respiratory syncytial virus (RSV) assays. One-button functionality and workflow flexibility allow the healthcare team to confidently use the device. In addition, the system’s fast and accurate results may help improve the patient experience. The BD Veritor™ Plus System streamlines the point-of-care testing experience.
The BD Veritor™ System SARS-CoV-2 & Flu A+B Assay require a BD Veritor™ Plus Analyzer with ROM Version 5.4 or Higher. A USB upgrade Key can be provided at no charge with order of test cassettes if needed.