Description
![]()
Contents: (1) Analyzer, (1) Handheld Power Supply Unit, (1) User Manual-English, (1) CD-ROM User Manual
Regulatory Status: CLIA Waived / Professional Use Only
Sample Type: Finger Stick Whole Blood
Determinations: Prothrombin Time (PT)/INR
Processing Time: 1 minute
Power Source: (4) AA Batteries or AC Adapter
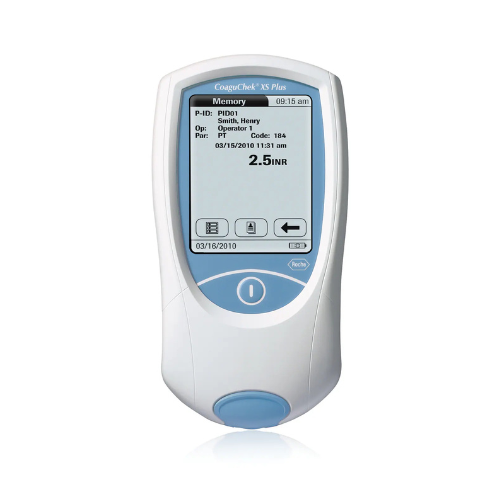
The CLIA-waived CoaguChek XS Plus system enables accurate PT/INR testing at the point of care. It also offers data management capabilities, so healthcare professionals can easily and efficiently manage warfarin patients at the point of care.
Test in any professional environment. Accurate and flexible technology provides results in about one minute and offers quality control options, including on-board controls and liquid quality controls.
Streamline data management. The CoaguChek XS Plus can store 2,000 test results and 500 optional quality control results. Additionally, it gives healthcare providers the ability to denote patient IDs and add comments.
Produce accurate results with ease. Only a small sample size is required, obtained through a fingerstick, and the icon-driven touch-screen interface makes testing a simple and intuitive process.
Features:
- It’s the only system with a 97% correlation to the lab using a single-strip platform, which means providers can count on consistent and accurate results.
- CoaguChek technology reduces heparin interference for more accurate results.
- The monitoring portfolio offers a single-strip platform for consistency in PT/INR results across the care continuum.
Resources: